|
We are about halfway through Lung Cancer Awareness Month and I would like to offer some information about a fantastic organization. If you are reading this, you likely know about this group. But, even if you do, I encourage you to sit back with your favorite beverage and take a few minutes to watch the video at the end of this post. When supporting any cause or charity with a financial gift, prudent questions are “Where does the money go?”, “How effective is the organization?”, “Is it worthy of my support?”, "What are they doing?", "What have they done?" I ask these questions myself before choosing to financially support any charitable cause. This video, narrated by their Senior Director of Patient Services and Programs, Danielle Hicks, does an excellent job of answering these questions for The Bonnie J. Addario Lung Cancer Foundation (ALCF). My family and I met the Addarios shortly after our mother died in 2007. We were immediately struck by their sincerity, warmth - and tenacity. But, we were also impressed by their team and how they were attacking the lung cancer problem with intelligence and professionalism. In 2010, The Joan Gaeta Lung Cancer Fund proudly became an affiliate of ALCF. And, since 2012, those of you in Georgia have been able to order Lung Cancer Awareness License Plates - a first in the United States. 85% of the annual tag fee goes to ALCF’s research institute. You can also donate directly to ALCF via this link. Please to watch the video and consider a donation during this important month. Thank you, Joseph A. Gaeta The Joan Gaeta Lung Cancer Fund
0 Comments
Please help us in our continuing effort..... Donate today. We cannot think of a better way to kick of Lung Cancer Awareness Month 2016 than with this excellent Q&A with a leader in the movement - and our friend - Bonnie Addario. Please read this inspirational and informative interview from Genentech: https://www.gene.com/stories/qa-bonnie-j-addario
The Bonnie J. Addario Lung Cancer Foundation has a full-page ad in the June issue of Delta Air Lines' Sky Magazine! Way to go, guys! Thanks for spreading the word and helping to save lives!
8/31/2015 TUNE IN TO "START-UP U" ON TUESDAY, SEPTEMBER 1ST! STARTUP LEGEND AND PREMIERE VENTURE CAPITALIST TIM DRAPER GENEROUSLY PLEDGES ONE MILLION DOLLARS TO CHARITY - INCLUDING THE ADDARIO LUNG CANCER FOUNDATION!!!Read NowDraper Will Donate the Money When Tuesday’s All-New Episode of “Start-Up U” Hits One Million ViewersBurbank, CA (August 18, 2015) – Start up legend Tim Draper will donate one million dollars to charity if as many viewers watch ABC Family’s unscripted series “Startup U” on Tuesday, September 1 at 10:01PM ET/PT on ABC Family.
On Saturday, Tim Draper announced via Twitter and Facebook that he will donate one million dollars to charity if one million viewers tune-in to Tuesday’s all-new episode. Not only does Draper have a long history of donating to worthy causes close to his heart, but he is a champion of entrepreneurism; his life’s mission is to create a better future for everyone. “I believe in the series and the great work these kids are doing. What better way to support them than connecting it back to a worthy cause? This is a win/win situation for everyone, but especially the charities.” Charities that will benefit from Draper’s benevolence are ones very close to his heart, including the Parkinson’s Institute (in honor of his mother, who fights this disease), Bizworld, Tourettes Association of America (his brother-in-law and nephews are affected), Smith College Women’s business plan competition, Endeavor (supports third world entrepreneurship), Defy (works with former prisoners when they have paid their debt to society, Cal Arts and California State Summer School for the Arts, and the Addario Lung Cancer Foundation (amember of his staff is a survivor). Viewership will be measured by all methods, including mobile and online viewing, L+3 and L+7. In Tuesday’s episode, titled “Bro’s & Bras,” it’s Evangelism Week, which teaches the students how to become experts at selling their brand. Tim putseveryone in yet another challenging situation when he sends them to San Francisco to sell men and women’s underwear to the many shoppers in Union Square. There’s a catch, though: they have to sell the undergarments based on only a picture, with no actual product. Frustrated by the process, Tony makes a decision that irritates Sequoia and his classmates. Sharon meets with resistance when she pitches Hotpoint to various venues around the Bay Area, only to have things get tougher when Tim puts her in the hot seat. Meanwhile, Michael Volpatt (Partner, Larkin/Volpatt Communications) and Charlie meet with several students to evaluate their brands. “Startup U” is produced by Ugly Brother Studios, a Sky Vision partner. Mike Duffy, Tim Duffy, Tim Draper, Perry Dance and Ted Iredell serve as executive producers. Part of the Disney|ABC Television Group, ABC Family is distributed in 94 millionhomes. ABC Family entertains and connects to audiences with bold, relatableprogramming that celebrates the epic adventure of becoming an adult, from first kiss to first kid. ABC Family's programming is a combination of network-defining original series and original movies, quality acquired series and blockbuster theatricals. “WATCH ABC Family” is an authenticated service which allows viewers with participating TV subscription services access to 24/7 live viewing of the network, as well as continued on-demand access to such popular series at home and on-the-go via a wide array of devices. ABC Family is also the destination for annual holiday events with "13 Nights of Halloween" and "25 Days of Christmas." ABC Family Media Relations: Joelle Johnson, (818) 569-7746, joelle.johnson@disney.com For additional press materials, please visit http://www.disneyabcpress.com/abcfamily/ Facebook: www.facebook.com/StartUpUTV Twitter: twitter.com/StartUpUTV 7/24/2015 The Bonnie J. Addario Lung Cancer Foundation Honors Leading Lung Cancer Researcher with the 2015 Addario Lectureship AwardRead NowFred R. Hirsch, MD, PhD, honored at the 16th International |
Details
|



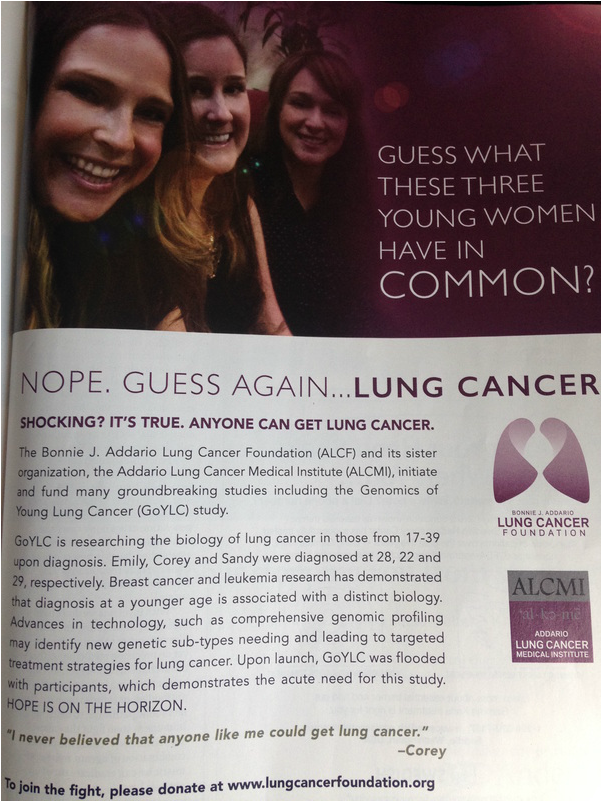





 RSS Feed
RSS Feed
